RHUBARB YELLOW
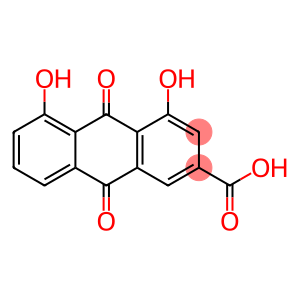
Rhein
CAS: 478-43-3
Molecular Formula: C15H8O6
RHUBARB YELLOW - Names and Identifiers
| Name | Rhein |
| Synonyms | Rhein RHEIN rheicacid monorhein RHUBARB YELLOW RHUBARB EXTRACT TIMTEC-BB SBB001152 chrysazin-3-carboxylicacid 1,8-Dihydroxy-3-Carboxy anthraquinone 1,8-DIHYDROXY-9,10-ANTHRAQUINONE-3-CARBOXYLIC ACID 4,5-dihydroxy-9,10-dioxo-9,10-dihydroanthracene-2-carboxylate 4,5-dihydroxy-9,10-dioxo-9,10-dihydroanthracene-2-carboxylic acid |
| CAS | 478-43-3 |
| EINECS | 207-521-4 |
| InChI | InChI=1/C15H8O6/c16-9-3-1-2-7-11(9)14(19)12-8(13(7)18)4-6(15(20)21)5-10(12)17/h1-5,16-17H,(H,20,21)/p-1 |
| InChIKey | FCDLCPWAQCPTKC-UHFFFAOYSA-N |
RHUBARB YELLOW - Physico-chemical Properties
| Molecular Formula | C15H8O6 |
| Molar Mass | 284.22 |
| Density | 1.3269 (rough estimate) |
| Melting Point | ≥300 °C (lit.) |
| Boling Point | 346.72°C (rough estimate) |
| Flash Point | 329.4°C |
| Water Solubility | <0.1 g/100 mL at 17 ºC |
| Solubility | Insoluble in water, soluble in pyridine, sodium bicarbonate aqueous solution, slightly soluble in ethanol, benzene, ammonia, ether and petroleum ether. |
| Vapor Presure | 3.85E-15mmHg at 25°C |
| Appearance | Yellow to orange powder |
| Color | Yellow to Dark Orange |
| Maximum wavelength(λmax) | ['432nm(MeOH)(lit.)'] |
| Merck | 14,8176 |
| BRN | 2222155 |
| pKa | 3.17±0.20(Predicted) |
| Storage Condition | 2-8°C |
| Stability | Hygroscopic |
| Refractive Index | 1.4413 (estimate) |
| MDL | MFCD00009618 |
| Physical and Chemical Properties | Rhizome of rhubarb derived from Polygonaceae |
| Use | Pharmaceutical Intermediates, health food raw materials |
RHUBARB YELLOW - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S37/39 - Wear suitable gloves and eye/face protection |
| WGK Germany | 2 |
| RTECS | CA9516000 |
| HS Code | 29189900 |
RHUBARB YELLOW - Reference Information
| Plant source: | Rhubarb |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| application | rhein has anti-tumor activity, antibacterial activity, immunosuppressive effect, diuretic effect, diarrhea effect, anti-inflammatory effect, treatment of diabetes and nephropathy effect. Health products are mainly used for reducing fat and weight, laxative and detoxification, cleaning the internal environment, preventing gastric cancer and delaying aging. |
| source | rhein has a wide range of sources. the rhizome of polygonaceae plant palm leaf rhubarb (RheumpalmatumL.), the root of polygonum multiflorum, the pod of legume plant sana angustifolia (Cassia angustifolia Vahl.), rutaceae plant ruta (Tuta;Grave-olensL.), and the root of Lilaceae plant. |
| pharmacological action | rhein contains chrysophanol, emodin, aloe emodin, rhein, sennoside and other chemical components. Both rhein and emodin have anti-tumor effects, especially have a strong inhibitory effect on melanoma, and also have a certain inhibitory effect on breast cancer and Ehrlich ascites cancer. Intratumoral administration of mouse breast cancer has obvious destructive effect on cancer tissues. The inhibition rates of 5 mg/kg rhein and emodin on mouse melanoma were 76% and 73% respectively. Emodin has a significant competitive inhibitory effect on tyrosinase, which may be one of the mechanisms of rhubarb's anti-melanoma action. Emodin concentration at 10 μg/ml can inhibit the division of human lung cancer A- 549 cells and significantly inhibit the DNA biosynthesis of lung cancer A- 549 cells. The crude extract of rhubarb was injected subcutaneously to inhibit sarcoma S37 in mice. The inhibition rate of rhein on Ehrlich ascites cancer was 15%, and that of mouse sarcoma S180 was 21%. The inhibition rate of hot water extract of rhubarb on mouse sarcoma S180 was 48.8%. Rhein has inhibitory effect on mouse leukemia P388. Rhein, emodin and aloe-emodin were extracted from rhubarb. These three anthraquinone derivatives all reduce the amount of ascites and the number of cancer cells in tumor mice to varying degrees. Among them, the effect of rhein is more obvious, and aloe-emodin is worse, which is basically parallel to prolonging survival. Rhein and emodin have strong inhibitory effects on DNA, RNA and protein biosynthesis, while aloe-emodin has weak inhibitory effects. |
| uses | pharmaceutical intermediates and health food raw materials have anti-inflammatory, anti-osteoarthritis and anti-cancer activities. It reduces the production and secretion of IL-1β, induces the activity of nitric oxide synthase, phosphorylation c-Jun and phosphorylation of c-Jun amino-terminal kinase (JNK). |
Last Update:2024-04-09 02:00:08
Supplier List
Product Name: Rhein Request for quotation
CAS: 478-43-3
Tel: 17505222756
Email: pules.cn@gmail.com
Mobile: +86-17551318830
CAS: 478-43-3
Tel: 17505222756
Email: pules.cn@gmail.com
Mobile: +86-17551318830
Spot supply
Product Name: Diacerein Impurity 4(Diacerein EP Impurity C) Visit Supplier Webpage Request for quotationCAS: 478-43-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Rhein Request for quotationCAS: 478-43-3
Tel: +86-17551318830
Email: r@reformchem.com
Mobile: +86-17551318830
QQ: 3785839865
Multiple SpecificationsSpot supply
Product Name: Rhein Visit Supplier Webpage Request for quotationCAS: 478-43-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Rhein Request for quotation
CAS: 478-43-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 478-43-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Spot supply
Product Name: Rhein Visit Supplier Webpage Request for quotationCAS: 478-43-3
Tel: +86-(0)731-84213302
Email: sales@staherb.cn
Mobile: +86 13875855783
QQ: 484702402
WhatsApp: +86 18374838656
Linkedin: +86 18374838656
Multiple SpecificationsSpot supply
Product Name: Rhein Visit Supplier Webpage Request for quotationCAS: 478-43-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Rhein Request for quotation
CAS: 478-43-3
Tel: 17505222756
Email: pules.cn@gmail.com
Mobile: +86-17551318830
CAS: 478-43-3
Tel: 17505222756
Email: pules.cn@gmail.com
Mobile: +86-17551318830
Spot supply
Product Name: Diacerein Impurity 4(Diacerein EP Impurity C) Visit Supplier Webpage Request for quotationCAS: 478-43-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Rhein Request for quotationCAS: 478-43-3
Tel: +86-17551318830
Email: r@reformchem.com
Mobile: +86-17551318830
QQ: 3785839865
Multiple SpecificationsSpot supply
Product Name: Rhein Visit Supplier Webpage Request for quotationCAS: 478-43-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Rhein Request for quotation
CAS: 478-43-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 478-43-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Spot supply
Product Name: Rhein Visit Supplier Webpage Request for quotationCAS: 478-43-3
Tel: +86-(0)731-84213302
Email: sales@staherb.cn
Mobile: +86 13875855783
QQ: 484702402
WhatsApp: +86 18374838656
Linkedin: +86 18374838656
Multiple SpecificationsSpot supply
Product Name: Rhein Visit Supplier Webpage Request for quotationCAS: 478-43-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History






